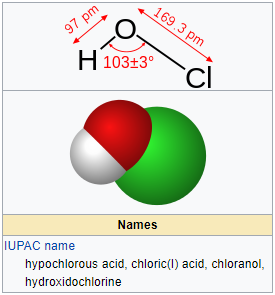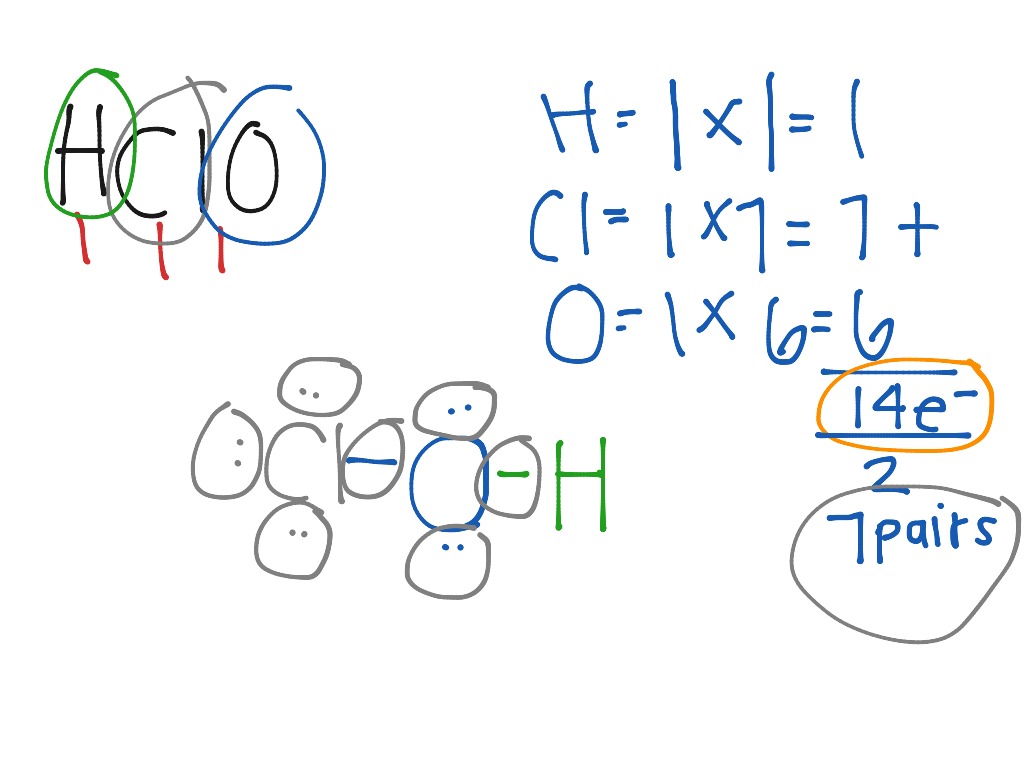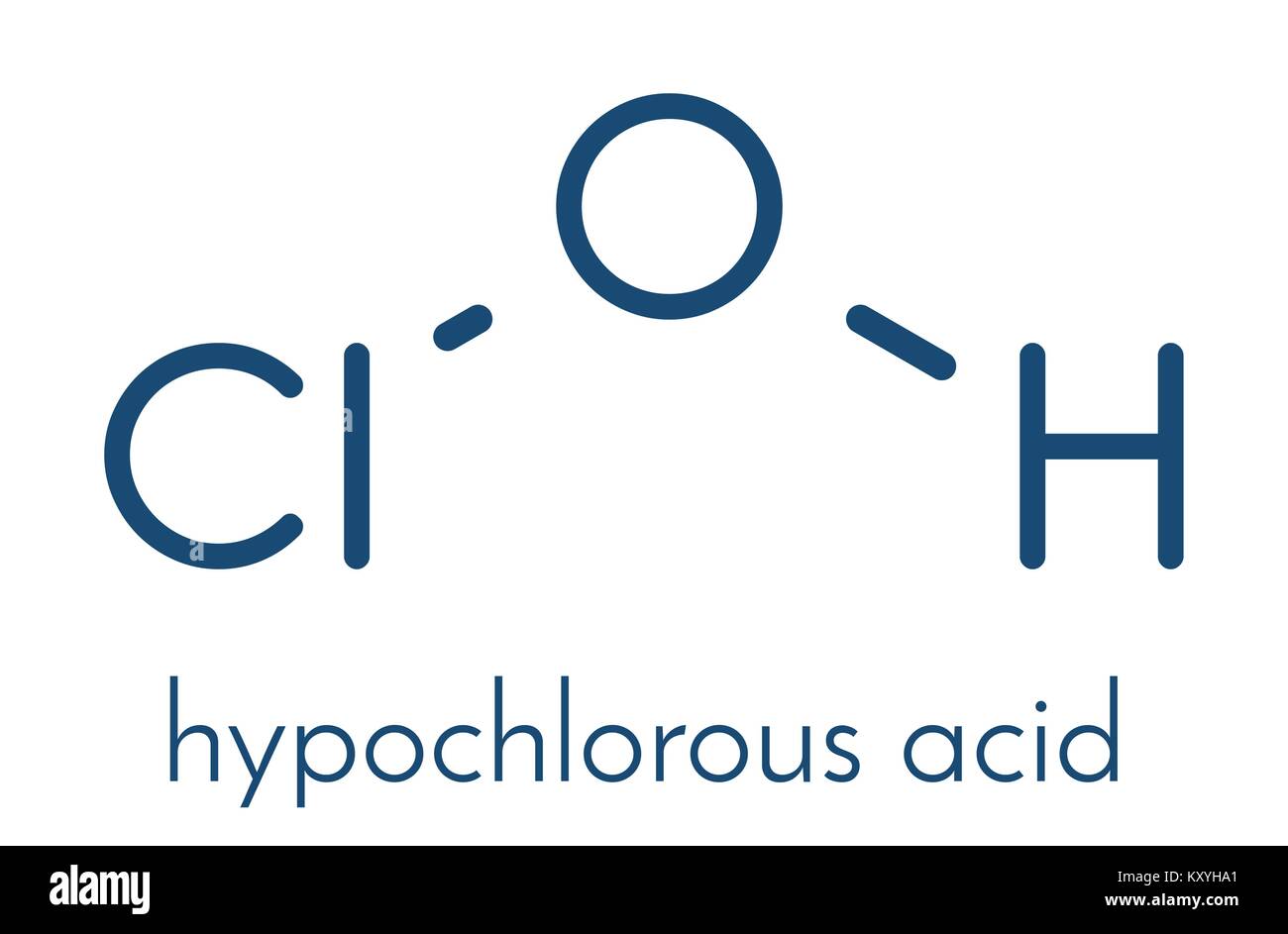
Hypochlorous acid (HClO) disinfectant molecule. Formed when chlorine is dissolved in water. Skeletal formula Stock Vector Image & Art - Alamy
pH profile of the concentrations of ClO -(), HClO (), and ClO -+ HClO () | Download Scientific Diagram
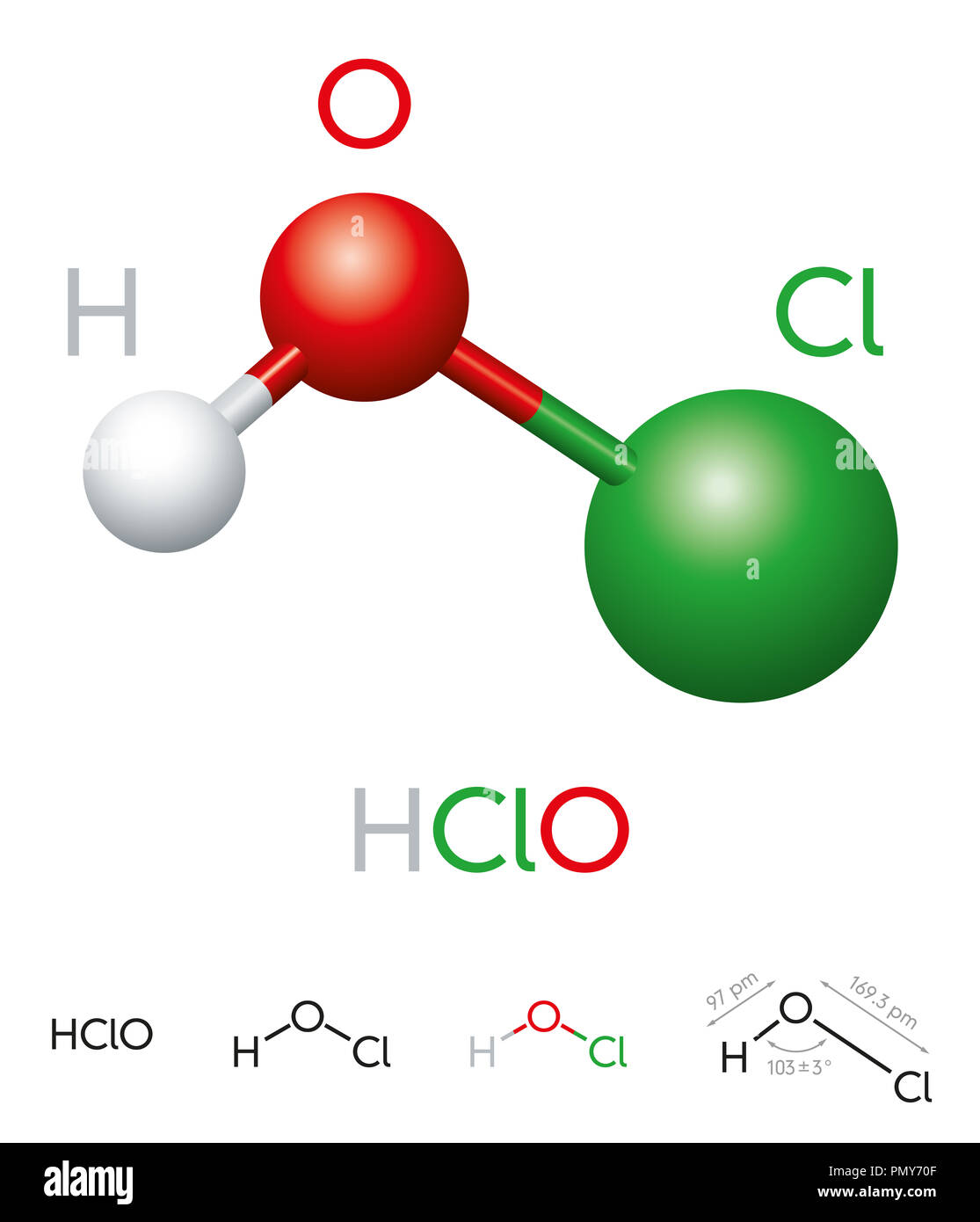
HClO. Hypochlorous acid. Molecule model, chemical formula, ball-and-stick model, geometric structure and structural formula. Weak acid. Illustration Stock Photo - Alamy

SOLVED: Hypochlorous acid (HClO) reacts with chloride ion to produce chlorine gas as follows: HClO(aq) + Cl-(aq) + H+(aq) → Cl2(g) + H2O(l) The measured cell potential under standard conditions for this
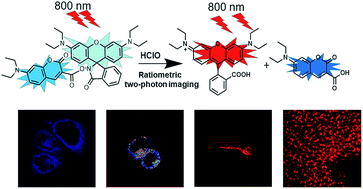
A ratiometric two-photon fluorescent probe for the rapid detection of HClO in living systems - Analytical Methods (RSC Publishing)

SOLVED: The Ka of hypochlorous acid (HClO) is 3.00 × 10-8. What is the pH at 25.0 °C of an aqueous solution that is 0.0200 M in HClO? +2.45 -2.45 -9.22 +9.22 +
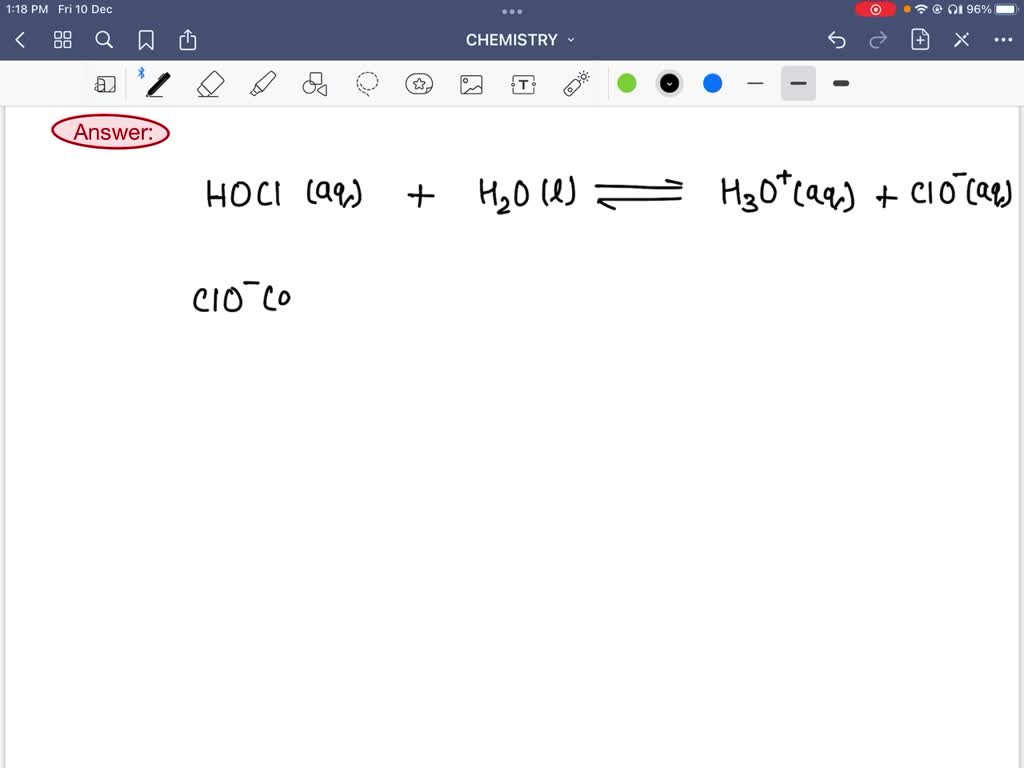
SOLVED: Hypochlorous acid ( HClO) is a weak acid. The conjugate base of this acid is the hypochlorite ion (ClO ). Wrtie a balanced equation showing the reaction of HClO with water.